Protein Crystal Mounting
Overview
The motivation for this project, along with some background information, is described separately.
Task Description

Fig.2: Crystal mounting tools

Video: A crystal mounting example (8MB)
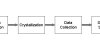
Fig.3: Crystal mounting in the HTP pipeline
The most-commonly used tool for mounting is the cryogenic loop (e.g. ones made by Hampton Research) though glass capillaries are also used. Recently, newly developed tools have been introduced, such as the micromounts manufactured by MiTeGen and our own microshovels. Fig.2 gives a visual comparison of the loop, the micromount and the microshovel.
The task is currently performed by skilled technicians. Mounting a crystal in a loop manually requires time, patience and excellent motor skills. Accuracy and speed are critical as the crystals are fragile and very sensitive to environmental changes. Dehydration quickly leads to crystal quality degradation. The video in the side panel is an example of what the task entails --- the loop in this video was installed on a micropositioner which was teleoperated. In reality, the task may be further complicated by the limited time for operation, "skins" forming on the surface of the droplet and crystals adhering to the coverslip.
Crystal mounting is a term that has been used to refer to both the task of picking up a protein crystal (a.k.a crystal harvesting) and the task of placing the tool with the crystal already on it on the beamline. In our work, we use the term to mean the former, and the latter we refer to as beamline mounting to avoid confusion (Fig.3).
System Design And Operation

Fig.4: Crystal mounting setup
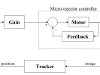
Fig.5: Control system block diagram

Video: Automatic crystal mounting (1MB)
The crystal mounting procedure starts with the placement of the necessary tools and objects in the work space (Fig.4). First, a microbridge with cryoprotector is placed at its designated location on the tray. Next, a micromount is installed on the fixture to the right and is positioned adequately so it is immersed in the cryprotector at an angle of approximately 45 degrees and is ready to receive the crystal. Finally, the user places a coverslip with the droplet containing the protein crystals on the microscope tray such that they are in the field of view.
The user starts the program and specifies which crystal (among the possibly many in the drop) is to be mounted. After that, the system proceeds autonomously: It immerses the pipette into the drop, approaches the crystal, aspirates the crystal, transitions from the drop on the coverslip to the cryoprotector in the microbridge and deposits the crystal in the mounting tool. Some of these steps are performed in open-loop fashion because the system is calibrated for the locations and dimensions of the relevant objects and the system actuators meet the requirement for positioning accuracy. The aspiration of the crystal, however, is an example of a step that crucially depends on reliable sensory feedback. To determine the location of the crystal and detect when the crystal is inside the pipette, we use region trackers applied to the visual feed from the camera. A control loop tracks the motion of the crystal as it is drawn into the pipette and adjusts the suction applied by the microinjector accordingly until the crystal is safely inside. A block diagram of the control algorithm is shown in Fig.5. A video of the system in operation is shown in the side panel.
Publications
-
Two-Stage Robotic Crystal Mounting of Protein Crystals
for X-Ray Data Collection
In Proc. of the IEEE Int. Conf. on Automation Science and Engineering (CASE'08), Arlington, VA, August 2008, pp.1019-1024 (paper & poster).
(with Peter K. Allen)
-
Visually-Guided Protein Crystal Manipulation Using
Micromachined Silicon Tools
In Proc. of the IEEE/RSJ Int. Conf. on Intelligent Robots and Systems (IROS'04), Sendai, Japan, September 2004, pp.236-241.
(with Peter K. Allen and William Edstrom)
-
Microrobotic Crystal Mounting Using Computer Vision
Microrobotics for Biomanipulation Workshop, IEEE/RSJ Int. Conf. on Intelligent Robots and Systems (IROS'03), Las Vegas, NV, October 2003.
(with Peter K. Allen and Youcef Mezouar)
-
Visual Servoed Micropositioning for Protein
Manipulation Tasks
In Proc. of the IEEE/RSJ Int. Conf. on Intelligent Robots and Systems (IROS'02), Lausanne, Switzerland, October 2002.
(by Youcef Mezouar and Peter Allen)


